Abstract
Heme (iron protoporphyrin IX) is a major toxic component of hemoglobin released from red cell turnover especially during intravascular hemolysis such as in sickle cell disease, malaria and paroxysmal nocturnal hemoglobinuria. Heme oxygenase 1 (HO-1) is an enzyme that degrades heme, primarily generated by damaged or senescent red blood cells, releasing iron from protoporphyrin and generating equimolar amounts of biliverdin and carbon monoxide. The HO-1 heme catabolic pathway provides an important defense from oxidant and inflammatory consequences of exposure to free heme. HO-1 induction occurs as a transcriptional response to heme through the NRF2 oxidant-sensing transcription factor. Placental Growth Factor (PGF) is an angiogenic growth factor secreted by proliferating erythroblasts during normal development, which we previously have found is also activated by heme, secondarily regulating expression of the vasoconstrictor endothelin-1 that promotes pulmonary hypertension. We assessed the transcriptional response of HO-1 and PGF in different organs in C57BL/6J wild type (WT) and Nrf2 null mice following acute exposure to heme.
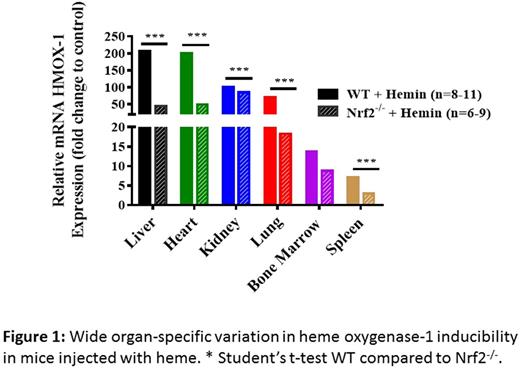
WT and Nrf2 null mice were injected intravenously with saline or hemin (120 µmoles/kg body weight), followed by harvest of the bone marrow, spleen, liver, kidney, lung and heart 3 hours after injection. Real time PCR was used to quantify transcripts and relative changes in expression. Heme oxygenase-1 mRNA levels rose sharply in organs from WT mice that received hemin compared with saline control: liver (207-fold), heart (201-fold), kidney (101-fold), lung (69-fold), bone marrow (13-fold) and spleen (7-fold). In Nrf2 null mice, induction of HO-1 mRNA expression by heme was blunted as expected, with the greatest HO-1 mRNA induction in the kidney, followed by the heart, liver, lung, bone marrow and spleen. Our findings suggest that PGF expression is Nrf2-dependent primarily in the bone marrow, lung and kidney (4-fold, 1.65-fold and 1.89-fold, respectively, WT compared to Nrf2 null). Interestingly, in some of the organs, we observed an inverse relationship between HO-1 inducibility and PGF expression in organs from Nrf2 null mice injected with hemin, suggesting that reduced clearance of heme promotes more robust PGF induction. We observed the highest basal expression of PGF (1.23 and 2.21 DeltaCt compared to GUSB) in the heart, rising even higher with heme exposure (0.51 and 0.80 DeltaCt) in WT and Nrf2 null mice respectively. This translates to 70% and 57% respectively of the level of the "housekeeping gene" GUSB, which constitutes relatively high-level expression. In WT mice injected with hemin, the heme importer Hrg1 mRNA induction was mild but significant (heart 2.5 fold, kidney 2.1-fold, and lung 2.2-fold), which are also the organs with highest HO-1 inducibility (all p<0.05). Hrg1 mRNA induction was strongly dependent on Nrf2. Hrg1 activation in the liver increased by 1.82-fold in WT mice injected with hemin (p=0.097).
The liver has been shown by others to take up free heme avidly, but these are the first data to demonstrate widely ranging differences in Nrf2-dependent heme response among other organs, implying their uptake of heme. We hypothesize that these differences in heme-induced HO-1 expression may reflect proportional differences in heme import, potentially via the Hrg1 heme importer that we find to be heme-inducible. There is likely also a role for the CD93 receptor for heme-hemopexin complexes and the FLVCR2 heme importer. This means that many organs may have some ability to take up free heme. Also, the ability of many organs to import extracellular heme might potentially play a role in diseases with robust release of heme into the circulation, such as β-thalassemia, sickle cell disease, malaria and other hemolytic anemias. PGF expression in the heart has previously shown by others to play a role in cardiac development and hypertrophy, and our new results point to cardiac PGF as a new hypothetical target of heme-induced dysregulation. Understanding the mechanism of organ-specific response to heme and PGF induction could expand the range of heme-mediated toxicity and pathophysiology of the heme-Nrf2-PGF-endothelin axis in sickle cell disease.
Ofori-Acquah: NuvOx Pharma: Patents & Royalties. Kato: Global Blood Therapeutics: Consultancy; Bayer: Research Funding; MAST Therapeutics: Research Funding; Novartis: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.